News and Announcements

Universal Biosensors Welcomes FDA Clearance for Xprecia Stride Analyzer
- Published October 06, 2016 12:03PM UTC
- Publisher Wholesale Investor
- Categories Company Updates
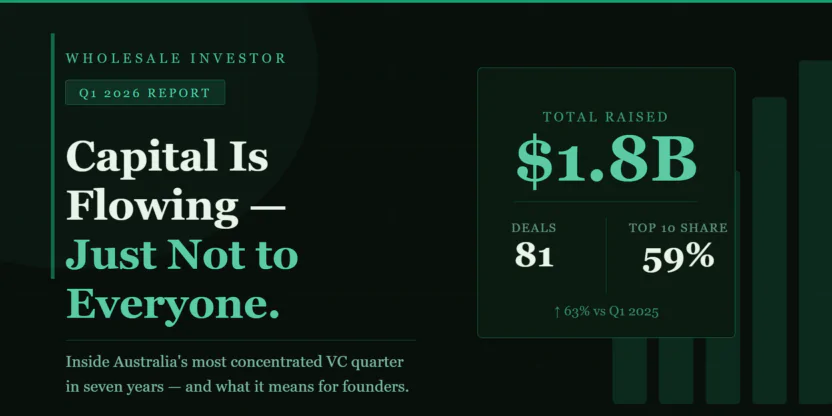
5th October 2016, ASX Announcement
Universal Biosensors (ASX: UBI) today welcomes news that the US Food and Drug Administration (FDA) has cleared for sale in the US the Xprecia Stride Coagulation Analyzer which is produced by partner Siemens Healthineers and uses test strips developed and manufactured by UBI.
The Xprecia Stride analyser is a handheld device which delivers Prothrombin time international normalised ratio testing for point-of-care monitoring and assessment of patients taking oral anticoagulation therapy warfarin.
To view the full announcement, please click on the button below.
Company Updates
Backed By Leading Investment Groups and Family Offices